 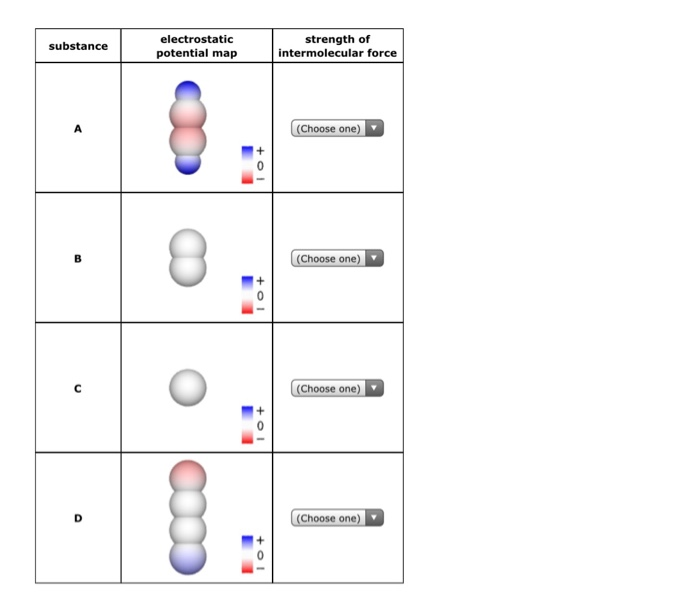
(For more information on the behavior of real gases and deviations from the ideal gas law, see Section 7.1.) These interactions become important for gases only at very high pressures, where they are responsible for the observed deviations from the ideal gas law at high pressures. Because electrostatic interactions fall off rapidly with increasing distance between molecules, intermolecular interactions are most important for solids and liquids, where the molecules are close together. Like covalent and ionic bonds, intermolecular interactions are the sum of both attractive and repulsive components. Intermolecular forces are electrostatic in nature that is, they arise from the interaction between positively and negatively charged species. Similarly, solids melt when the molecules acquire enough thermal energy to overcome the intermolecular forces that lock them into place in the solid. Liquids boil when the molecules have enough thermal energy to overcome the intermolecular attractive forces that hold them together, thereby forming bubbles of vapor within the liquid. Intermolecular forces determine bulk properties such as the melting points of solids and the boiling points of liquids. 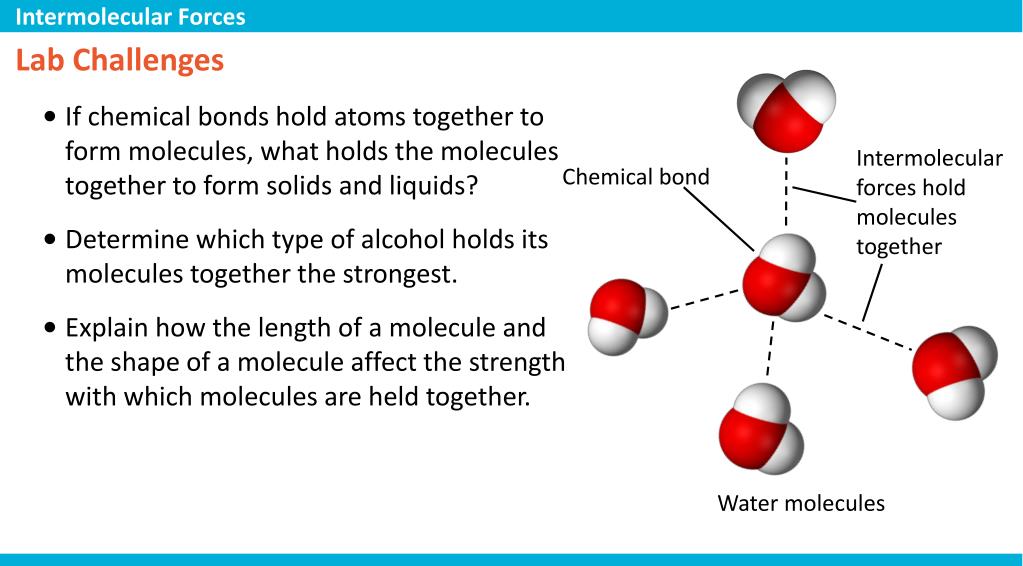 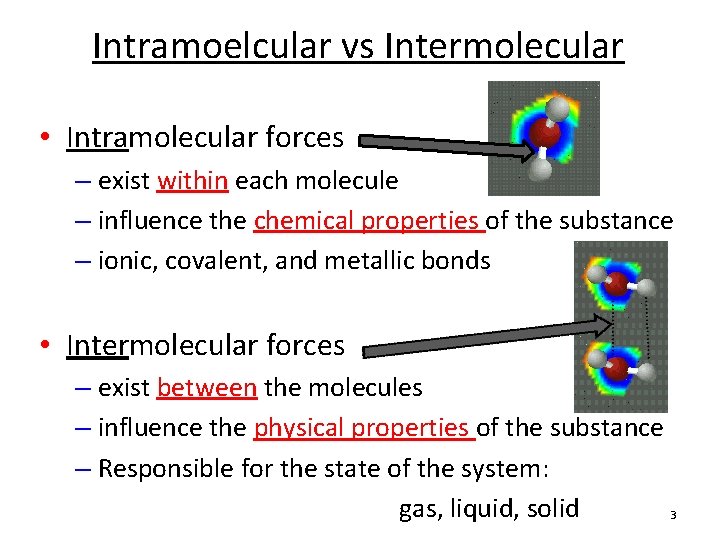
The properties of liquids are intermediate between those of gases and solids but are more similar to solids. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
